Guanidinylation
By introducing guanidine groups into molecules, guanidylation can significantly alter the chemical and biological properties of molecules. It is often used in drug synthesis, catalytic reactions or biomolecular modification.
Technical Resources
-
1
 1
Catalytic asymmetric Friedel–Crafts alkylation of unprotected indoles with nitroalkenes using a novel chiral
1
Catalytic asymmetric Friedel–Crafts alkylation of unprotected indoles with nitroalkenes using a novel chiralChiral chloro-indeno pybox has served as a new ligand for the Yb(OTf)3-catalyzed asymmetric Friedel–Crafts alkylation reaction of indoles with nitroalkenes. The tunable nature of pybox ligands
-
2Catalytic asymmetric Friedel–Crafts alkylation of unprotected indoles with nitroalkenes using a novel chiral
-
3Catalytic asymmetric Friedel–Crafts alkylation of unprotected indoles with nitroalkenes using a novel chiral
-
4Catalytic asymmetric Friedel–Crafts alkylation of unprotected indoles with nitroalkenes using a novel chiral
-
5Catalytic asymmetric Friedel–Crafts alkylation of unprotected indoles with nitroalkenes using a novel chiral
-
In-stockPurity: ≥95%
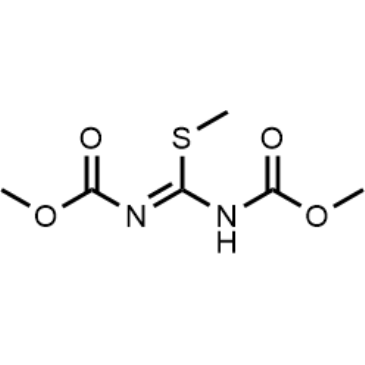 Cat. No. : CS-0112935CAS No. : 34840-23-8
Cat. No. : CS-0112935CAS No. : 34840-23-8* This product is not sold in the region.
-
In-stockPurity: ≥97%
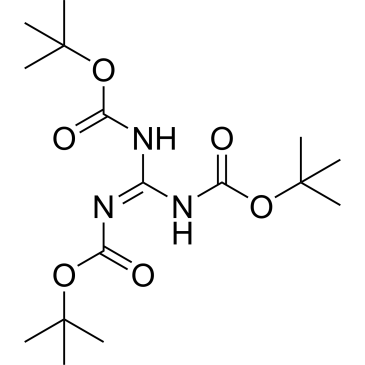 Cat. No. : CS-0173626
Cat. No. : CS-0173626 -
In-stockPurity: ≥95%
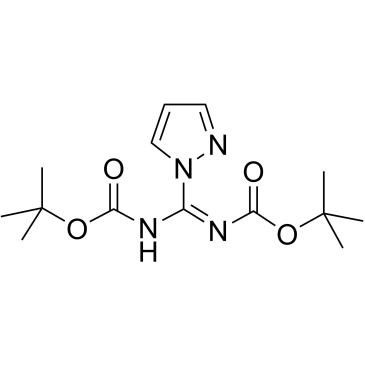 Cat. No. : CS-W002736CAS No. : 152120-54-2
Cat. No. : CS-W002736CAS No. : 152120-54-2* This product is not sold in the region.
-
In-stockPurity: ≥98%
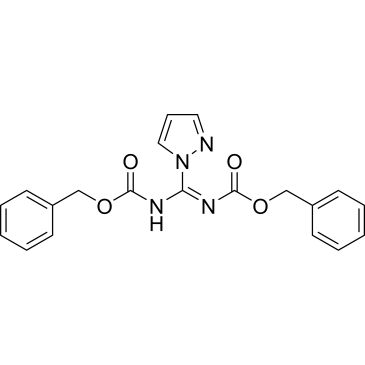 Cat. No. : CS-W008067
Cat. No. : CS-W008067 -
In-stockPurity: ≥96%
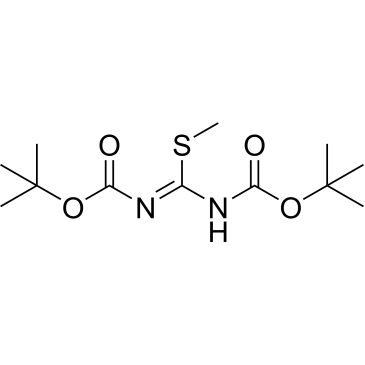 Cat. No. : CS-W008142CAS No. : 107819-90-9
Cat. No. : CS-W008142CAS No. : 107819-90-9* This product is not sold in the region.
-
In-stockPurity: ≥98%
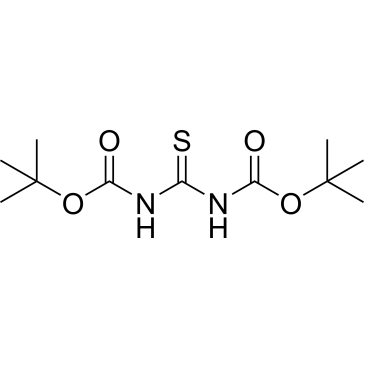 Cat. No. : CS-W014572CAS No. : 145013-05-4
Cat. No. : CS-W014572CAS No. : 145013-05-4* This product is not sold in the region.
-
In-stockPurity: ≥97%
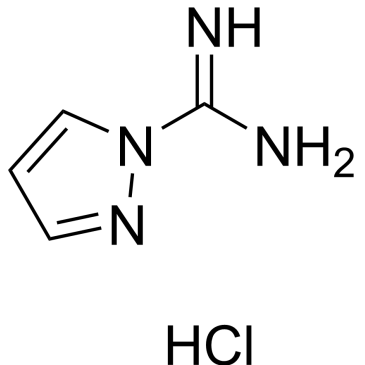 Cat. No. : CS-W017748CAS No. : 4023-02-3
Cat. No. : CS-W017748CAS No. : 4023-02-3* This product is not sold in the region.
-
In-stockPurity: ≥97%
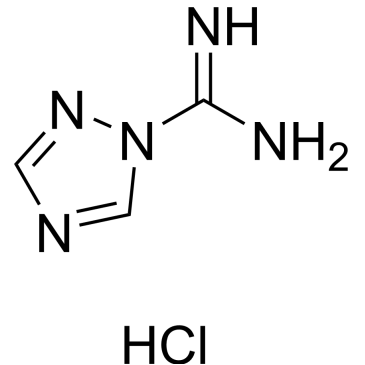 Cat. No. : CS-W018199CAS No. : 19503-26-5
Cat. No. : CS-W018199CAS No. : 19503-26-5* This product is not sold in the region.
-
In-stockPurity: ≥97%
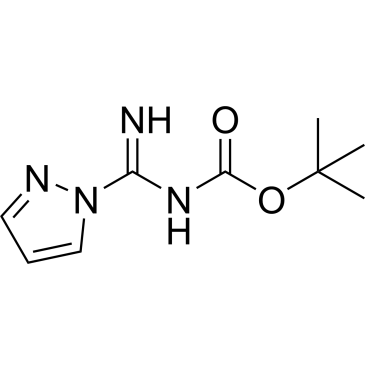 Cat. No. : CS-W020385
Cat. No. : CS-W020385 -
In-stockPurity: ≥98%
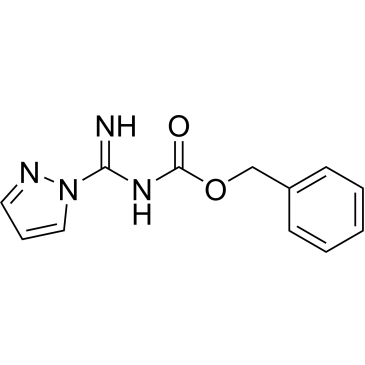 Cat. No. : CS-0150031CAS No. : 152120-62-2
Cat. No. : CS-0150031CAS No. : 152120-62-2* This product is not sold in the region.


